|
12/7/2023 0 Comments Is sf2 linearWhen carbon forms single bonds with other atoms, the shape is tetrahedral. It all has to do with its atomic orbital hybridization. Is CO2 trigonal pyramidal?ĬO2 is not trigonal planar, but is linear (O = C = O). In the carbon dioxide, the two double bonds try to get as far apart as possible, and so the molecule is linear. 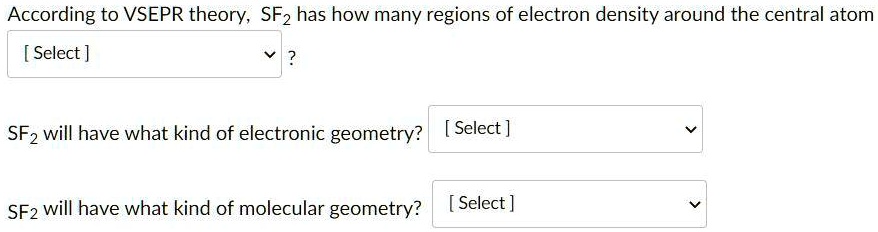
Carbon dioxide is linear, while sulphur dioxide is bent (V-shaped).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
